INTRODUCTION
Worldwide, tobacco smoking causes the death of around 8 million people every year1. In Italy, more than 70000 deaths, corresponding to more than 12% of total mortality, are annually attributable to smoking2. The large cohort of British male doctors followed for 50 years, between 1951 and 2001, showed that the life expectancy of smokers was 10 years less compared to that of lifelong non-smokers. Also, this study was able to highlight the beneficial effects of smoking cessation estimating the extent of the reduction in mortality when cigarette smoking is stopped at different ages. In particular, it has been shown that subjects who had quit at age 30 or 40 years had practically the same life expectancy as never smokers. Also, quitting at age 60 or 50 years resulted in a gain of about 3 and 6 years of life expectancy, respectively3.
In Italy, in 2017–2019, the prevalence of current smokers among adults was around 22% (27% in men and 19% in women) and that of former smokers was approximately 13%4. The large majority of Italian former smokers had quit without any help5, but today there are pharmacological and psychological supports that greatly increase the rate of success in smoking cessation6. In Italy, the National Health Service (NHS) provides these supports to the general population through a number of healthcare providers, mainly pneumologists or psychologists or counsellors and nurses, located in around 300 smoking cessation services (SCSs) of northern, central and southern Italy7. In these SCSs, first line medications include varenicline and nicotine replacement therapies (NRT). In addition, individual and group psychological therapies have been shown to increase quit rates6.
The possibility to accurately predict the rates of success for a new incoming smoker willing to quit, would allow clinicians of SCSs to best propose a personalized therapeutic course. Accordingly, the international smoking cessation guidelines foresee different therapeutic courses depending by the patient readiness and motivation to quit8. For this reason, tools able to predict absolute rates of success for new patients of SCSs would be extremely important to improve their chances of a successful smoking cessation.
Selected sociodemographic and smoking-related characteristics are important determinants of smoking cessation. Thus, advanced age9, high level of education, as well as low nicotine dependence10-12 and low number of cigarettes smoked per day11,13 have all been shown to be directly associated with increased cessation success. Regarding sex/gender, contrasting results have been found in the success of quitting smoking among men and women, with men more likely to maintain long-term abstinence than women14. A psychological variable that has been also considered in the evaluation of patient’s process towards smoking cessation is the self-efficacy, that is the awareness of being able to carry out specific activities or to handle situations or aspects of one’s psychological or social functioning to complete a task successfully15. In this case it refers to the cognition of patients to be able to quit smoking. Some studies showed that self-efficacy might represent a relatively reliable predictor of smoking cessation15-17.
The main objective of this work is to evaluate the main predictors of the successful outcome of smoking cessation attempts in a group of smokers attending the SCS of Careggi Hospital in Florence (Italy) to quit smoking. Since from 2018 the information of smokers’ self-efficacy was added to the items collected at the first visit at the SCS, we analyzed the role of self-efficacy in predicting smoking cessation to verify whether the already existing evidence on the issue was further confirmed in this set of Italian smokers.
METHODS
We collected data of 576 consecutive current smokers attending for the first time the SCS of Careggi Hospital in Florence (Italy) between January 2018 and December 2019, with the aim of quitting smoking. Overall, 96 subjects who attended only the first visit (without formally starting the course of treatment) and 2 subjects who had a missing value for self-efficacy (i.e. the main outcome of this study) were excluded. Therefore, for the present analysis we considered 478 patients (224 men and 254 women) aged 15–83 years (mean: 54 years). Patients agreed to participate in the study and signed an informed consent form adopted by the Careggi outpatient service.
During the first visit, smokers were asked to provide information on various sociodemographic characteristics, such as sex, age, education level, and marital status. Also data on smoking-related variables were collected and these included smoking intensity (number of cigarettes smoked per day), number of pack-years (number of 20-cigarette packs smoked per day multiplied by the number of years of smoking), number of previous cessation attempts, and the Fagerström test for nicotine dependence (FTND) score. The FTND consists of six questions to establish the degree of physical dependence on nicotine, categorized as ‘low’ (0–4), ‘medium’ (5–7) and ‘high’ (8–10)18. Moreover, the healthcare provider measured the concentration (ppm) of carbon monoxide (CO) exhaled. The treating clinician also collected variables related to smoking cessation support and clinical conditions, including waiting time, the physician delivering the treatment, the participation to the motivational group (i.e. a weekly group moderated by professional educators, whose participation is on voluntary basis, proposed in addition to the individual intervention and the agreed therapy), the agreed treatment among pharmacological therapy (i.e. NRT, zyban, varenicline, cytisine), electronic cigarette, or behavioral therapy only (all the patients received a behavioral therapy together with the pharmacological intervention or the electronic cigarette), origin (i.e. whether the subject went to the SCS willingly or if he/she was encouraged by a healthcare professional), and concomitant diseases of the patient. Baseline sociodemographic and smoking characteristics of the 478 patients included in this study are described in Supplementary file Table 1.
Table 1
Distribution of patients to evaluate the association between patients’ characteristics and the probability of continuous smoking abstinence at different time evaluations and overall, Careggi Hospital, Florence, Italy, 2018–2019 (N=478)
| Variable | n (%) | Time | Overall | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 3 months | 6 months | 12 months | Univariable analysisb | Multivariable analysisc | ||||||
| % success | RR (95% CI) | % success | RR (95% CI) | % success | RR (95% CI) | p heterogeneitya | RR (95% CI) | RR (95 % CI ) | ||
| All subjects | 478 (100) | 47.9 | - | 40.2 | - | 33.9 | - | - | - | - |
| Sex | ||||||||||
| Male (Ref.) | 224 (46.9) | 44.6 | 1 | 38.8 | 1 | 32.6 | 1 | - | 1 | 1 |
| Female | 254 (53.1) | 50.8 | 1.14 (0.94–1.38) | 41.3 | 1.06 (0.85–1.33) | 35.0 | 1.08 (0.84–1.38) | 0.501 | 1.14 (0.94–1.38) | 1.17 (0.98–1.40) |
| Age (years) | ||||||||||
| <50 (Ref.) | 153 (32.0) | 40.5 | 1 | 35.3 | 1 | 28.8 | 1 | - | 1 | 1 |
| 50–60 | 170 (35.6) | 49.4 | 1.22 (0.95–1.56) | 40.0 | 1.13 (0.85–1.50) | 35.9 | 1.25 (0.91–1.72) | 0.891 | 1.22 (0.96–1.56) | 1.22 (0.96–1.54) |
| >60 | 155 (32.4) | 53.5 | 1.32 (1.04–1.68) | 45.2 | 1.28 (0.97–1.69) | 36.8 | 1.28 (0.93–1.77) | 0.980 | 1.32 (1.04–1.68) | 1.35 (1.07–1.71) |
| Fagerström score | ||||||||||
| 0–4 (Ref.) | 129 (27.0) | 49.6 | 1 | 41.9 | 1 | 35.7 | 1 | - | 1 | 1 |
| 5–7 | 236 (49.4) | 51.7 | 1.04 (0.84–1.29) | 42.4 | 1.01 (0.79–1.30) | 37.7 | 1.06 (0.80–1.40) | 0.973 | 1.04 (0.84–1.29) | 1.07 (0.87–1.30) |
| 8–10 | 113 (23.6) | 38.1 | 0.77 (0.57–1.03) | 33.6 | 0.80 (0.58–1.12) | 23.9 | 0.67 (0.45–1.00) | 0.785 | 0.76 (0.57–1.02) | 0.78 (0.58–1.05) |
| Self-efficacy score | ||||||||||
| 1–5 (Ref.) | 139 (29.1) | 36.0 | 1 | 28.8 | 1 | 22.3 | 1 | - | 1 | 1 |
| 6–7 | 152 (31.8) | 47.4 | 1.32 (1.00–1.74) | 40.1 | 1.39 (1.01–1.93) | 34.9 | 1.56 (1.07–2.28) | 0.774 | 1.33 (1.01–1.76) | 1.40 (1.06–1.85) |
| 8–10 | 187 (39.1) | 57.2 | 1.59 (1.23–2.05) | 48.7 | 1.69 (1.25–2.28) | 41.7 | 1.87 (1.31–2.66) | 0.776 | 1.60 (1.24–2.07) | 1.64 (1.28–2.12) |
a The null hypothesis of homogeneity among RRs calculated at different months for each variable of interest was tested using a log-binomial model, including the variable, time, and the interaction between time and variable as covariates. The p-value associated with the interaction parameter was reported.
b RRs were estimated, for each variable of interest, using log-binomial models, including the variable and time as covariates. To account for correlation within subjects, the generalized estimating equations (GEE) method was used. A first-order autoregressive working correlation structure was specified in the model.
c RRs were estimated using a log-binomial model, including all the variables reported in the table and time as covariates. To account for correlation within subjects, the generalized estimating equations (GEE) method was used. A first-order autoregressive working correlation structure was specified in the model.
The self-efficacy in predicting the outcome of cessation attempt was collected during the first visit. This was the self-perception of success of the cessation attempt by the current smokers and was measured on a 1–10 discrete scale ranging from 1, indicating ‘no hope of success’, to 10, indicating ‘confidence of success’.
For each participant, continuous abstinence was assessed through three follow-up visits at 3, 6 and 12 months. During each visit the concentration (ppm) of CO exhaled was recorded and self-reported smoking status and smoking intensity were collected. Self-reported smoking status was assessed at each follow-up and validated through the CO exhaled measurements. Subjects defining themselves as non-smokers were confirmed to be non-smokers if the exhaled CO was ≤6 ppm. Those few subjects defining themselves as non-smokers but with the exhaled CO ≥7 ppm were classified as smokers. Subjects lost at follow-up (165 at 3 months; 204 at 6 months; and 287 at 12 months) were considered to have remained smokers at the corresponding time.
Statistical analysis
Log-binomial models for longitudinal data were run to evaluate the association between patient’s characteristics and the probability of success of smoking cessation overall and at the different times of evaluation (i.e. 3, 6, and 12 months). To account for correlation within subjects, the generalized estimating equations (GEE) method was used. A firstorder autoregressive working correlation structure was specified in the model. We derived relative risks (RRs) and corresponding 95% confidence intervals (CIs) after adjustment for sex, age, FTND score, and self-efficacy. Self-efficacy, age, and FTND score were considered in approximate tertiles.
Statistical analyses were conducted with the SAS version 9.4 statistical package (SAS Institute, Cary, NC, USA). The statistical significance level was set at a two-tailed p<0.05.
RESULTS
Of the 478 current smokers, 229 participants (47.9%) were non-smokers at 3 months follow-up (44.6% of men and 50.8% of women), 192 (40.2%) continued abstinence at 6 months (33.8% of men and 41.3% of women) and 162 (33.9%) at 12 months (32.6% of men and 35.0% of women).
Figure 1 shows the percentage of subjects who successfully quit smoking at various times of evaluation according to self-efficacy score. Smoking cessation was higher among subjects with higher scores of self-efficacy and decreased with increasing time of follow-up.
Figure 1
Observed percentages of smoking cessation by time and self-efficacy category, Careggi Hospital, Florence, Italy, 2018–2019 (N=478)
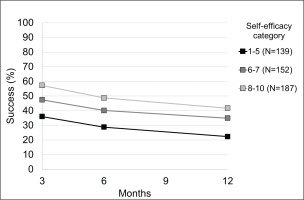
Table 1 shows the association between patients’ characteristics and the probability of success in smoking cessation at different times of evaluation and overall. Older subjects were more likely to successfully quit smoking compared to younger ones (the multivariable RR for >60 vs <50 years was 1.35; 95% CI: 1.07–1.71). Self-efficacy was directly associated with a success in smoking cessation, the multivariable RR for high (8–10) versus low (1–5) self-efficacy being 1.64 (95% CI: 1.28–2.12). Sex and FTND were not significantly associated with smoking cessation: the multivariable RR for sex (women vs men) was 1.17 (95% CI: 0.98–1.40) and that for high (8–10) vs low (0–4) FTND was 0.78 (95% CI: 0.58–1.05). The adjustment for education level, the delivered treatment, group therapy, and the number of prior quit attempts did not change the multivariable RRs for self-efficacy.
DISCUSSION
Our study found that self-efficacy represents an independent determinant of smoking cessation in smokers undergoing a cessation program at one SCS in Italy. This result is in broad agreement with those from other studies conducted on the issue. For example, a study based on 600 African American smokers revealed that smokers who successfully quit after six months of follow-up had higher baseline levels of self-efficacy compared to smokers who did not quit smoking16. Another study carried out in Switzerland on 115 patients showed that self-efficacy scores in smokers predicted smoking abstinence after 16 months of follow-up19. More importantly, a meta-analysis published in 2009, based on 54 prospective studies, suggested that the confidence in the ability to abstain was a consistent predictor of smoking cessation outcomes20.
The same meta-analysis, however, showed that the association between self-efficacy and future abstinence is less robust than expected20. Accordingly, in our study more than 20% of those with a low baseline level of self-efficacy successfully quit after one year. Moreover, almost 60% of those with a strong confidence to quit failed to be abstinent after 12 months. Although our study did not collect information on motivation and intention to quit, the latter finding might be explained in terms of a relatively limited motivation to quit in these subjects. Indeed, among motivated people, self-efficacy has been shown to play an extremely important role in predicting cessation21. It is also possible that those with a strong confidence to quit might refute pharmacological or psychological supports, thus possibly engaging in more high-risk situations22.
In our study, among the potential determinants considered, self-efficacy was the strongest predictor of successful smoking cessation. We found a weaker – although statistically significant – direct association between age and smoking cessation. Moreover, in contrast with other studies23, we did not find a statistically significant association with smoking cessation for sex or nicotine dependence. A recent study found that having had at least one quit attempt in the prior year was directly associated with quitting self-efficacy24. However, even after adjusting the model for the number of prior quit attempts, the main results on the association between self-efficacy and quit success did not substantially change, thus we were not able to demonstrate that the predictive power of self-efficacy is mediated by quit history.
Limitations
This study has some limitations, including the relatively limited sample size from one single SCS. Moreover, we assessed self-efficacy through one single question before treatment, and not through a validated scale. In addition, the study questionnaire assessed only self-efficacy as psychosocial variable, while other relevant variables, such as attitudes, social norms, motivation and intentions to quit, were not collected. Also, information about the rejected treatment supports was not available. Since these variables are all associated with self-efficacy, the effect of self-efficacy on smoking cessation observed in the study might be partly explained by other unmeasured variables. Finally, a power calculation for the considered sample size was not conducted a priori, but all consecutive patients going to the smoking cessation service were included. The study strengths include the prospective study design with a follow-up at 12 months and the validated assessment of smoking status at follow-up, through the measurement of the exhaled CO of all the study participants.
CONCLUSIONS
In our prospective study conducted in Italy, selfefficacy represents an independent and significant determinant of tobacco abstinence among smokers attempting to quit. We recommend to systematically collect self-efficacy among smokers undergoing a smoking cessation program in a SCS. This variable should be added to sociodemographic and smoking-related characteristics, and motivation to quit, to highly improve prediction models capable of providing a success rate for individual patients attempting to quit smoking25. Moreover, healthcare providers should implement strategies aimed at developing and maintaining high levels of self-efficacy and to further motivate smokers with a weak confidence to quit. This can be done by practicing coping strategies for craving and other cessation symptoms, setting achievable goals, and obtaining support from family, friends, or a support group. Increasing self-efficacy and confidence to quit could have major implications from a public health perspective, since there is evidence that these factors are able to increase quit success. Moreover, identifying factors that predict success contributes to improving treatment and moving towards a personalized care for smoking cessation26.


